|
Since 1980s to till date, chemiosmosis or proton-centric coupling became the mainstay explanation for mOxPhos in leading reviews 16, 17 and textbooks. These explanations also see a trans-membrane phenomenon as the primary drive for ATP-synthesis. 12 Some other hypotheses were also advocated: (1) Lehninger-Kasumov proposed a mechano-chemiosmotic model which posits that Complex V serves as a Ca 2+/H +–K +Cl – pump 13 and (2) Nath’s torsion mechanism 14, 15 posits that Complex V derives energy for phosphate-coupling by a trans-membrane anion/counter-cation gradient/exchange. Such developments inspired Paul Boyer to propose that re-entry of protons into the matrix (via the F o module) gave the rotary synthesis of ATP by F 1 module of Complex V. He proposed that a “proton-surplus” thus formed within the inter-membrane space leads to a pmf or trans-membrane proton potential (TMP) that drives ATP-synthesis within the matrix. Mitchell had postulated that the ETC components served to pump protons into the inter-membrane space, across the inner mitochondrial membrane. 7 - 11 Also, the chemiosmosis proposal could forge a connection between the electron transport chain (ETC, a medley of membrane-bound redox-active protein assemblies and diffusible smaller molecules) and F oF 1ATPase (or Complex V, the “coupling factor”). 5 Long-drawn efforts to identify any enzyme-phosphorylating intermediates failed (including Paul Boyer’s phased-out proposal of “phosphohistidine”), 6 and the proton-gradient hypothesis gained grounds. 4 Another group led by Peter Mitchell rooted for a “proton-motive force (pmf)” driving the ATP-synthesis. One school of thought with Edward Slater as the key exponent advocated an “enzyme-bound high-energy phosphorylating intermediate”. The era from 1950 to 1975 saw the research community divided on the mechanism of mOxPhos, the signature metabolic activity of mitochondria. This “oxidation of reducing equivalents using molecular oxygen, coupled with the synthesis of ATP in mitochondria” is the formal definition of mOxPhos. In the overall process, some reduced molecules (like reduced nicotinamide adenine dinucleotide (NADH)/flavin adenine dinucleotide (FADH 2)) are formed, which are oxidized using molecular oxygen at the inner mitochondrial membrane to yield ATP within the organelle’s matrix. 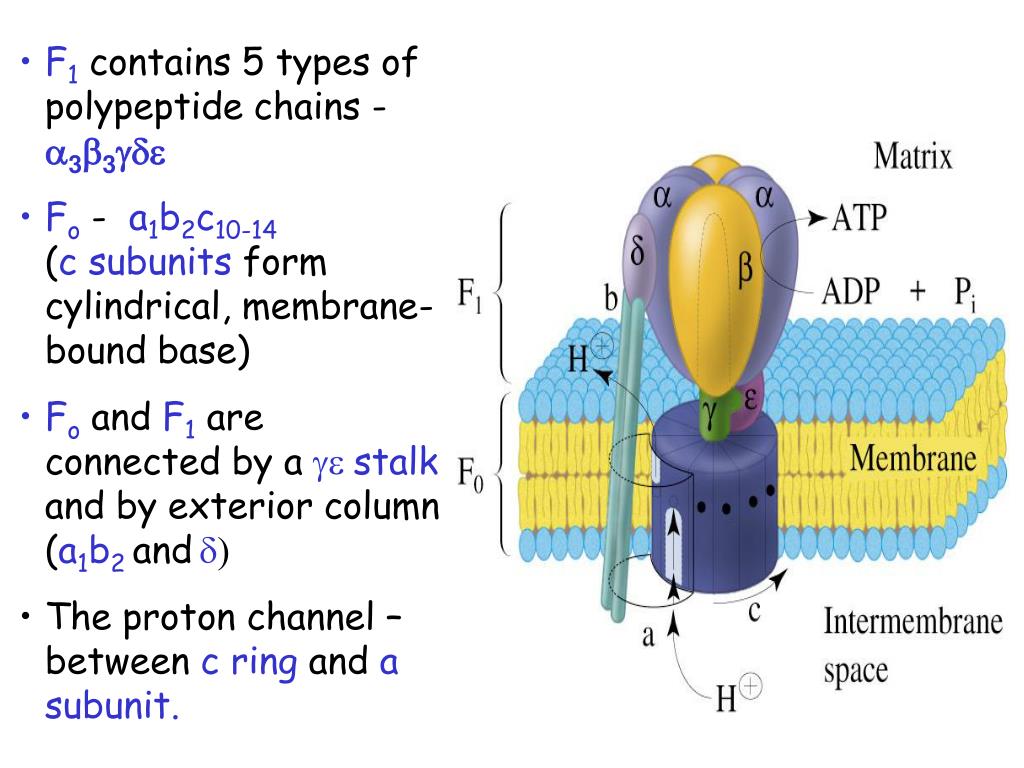 
In turn, this acetyl group is taken up by mitochondrial Krebs’ cycle, churning out two fully oxidized molecules of CO2. As a pre-requisite to mOxPhos, a systematic trimming and gradual stripping of electrons (or hydrogen atoms) of/from the multi-carbon reduced molecule (as exemplified by glucose, fatty/amino acids, etc) occurs to give a 2-carbon acetyl moiety. Cellular respiration is the metabolic routine that makes adenosine triphosphate (ATP the ubiquitous storable and transportable chemical currency) and is technically called mitochondrial oxidative phosphorylation (mOxPhos). 1 - 3 At present called “the powerhouses” of aerobic eukaryotes, mitochondria are deemed as the center-stage for generating the major chunk of chemical energy required for performing the “works” of life. By mid-20th century, the efforts of several researchers and pioneers such as Hans Krebs, George Palade, Fritiof Sjostrand, Britton Chance, and Albert Lehninger had raised the awareness on the then mysterious sub-cellular organelles. A Survey of the Various Views on Aerobic Cellular RespirationĮrroneous perceptions prevailed at the dawn of 20th century that mitochondria, “minute observations in microscopy”, were parasitic infections of cells.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
